Tasso+ Kit
Europe
The Tasso+ Kit includes a Tasso+ device, an IVD collection tube, and additional collection materials to provide user convenience such as alcohol swabs and bandages. The kit is intended for prescription use. The compatible tubes enable specific diagnostic use depending on the tube utilized in the kit and its intended use.
Tasso+ Kits are procedure packs per Article 22 of the MDR (EU) 2017/745. The Tasso+ device is CE marked and MHRA registered. For applications within the United States, please see our U.S. Tasso+ Kit page.
Tasso+ Kit
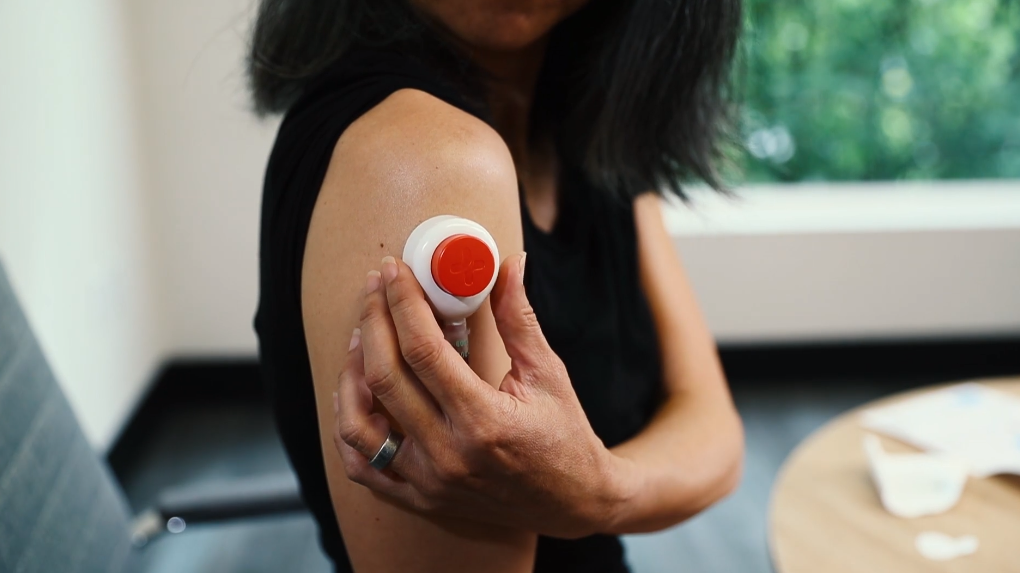
Tasso+ Kit In Use
Tasso+ Kit Video Instructions

Tasso+ Kit Details

Kit Intended Purpose: Allows self-collection of a capillary blood sample in conjunction with a compatible sample collection reservoir that is shipped to an accredited laboratory for analysis.
Kit Size and Weight: 180x157x38mm (7.1x6.2x1.5in), 178g (6.25oz)
Shipping: Sample can be shipped using packaging materials provided with the kit.
Kit Storage: Kit Storage: 18-25° C (64.4-77° F)
Kit Transport: Transport at 25-85% RH
Shelf Life: Up to 18 months, dependent on kit components. Tasso+ device is 24 months.
Manufacturing: Assembled in the USA
Availability: Tasso+ Kits are procedure packs per Article 22 of the MDR (EU) 2017/745. The Tasso+ device is CE marked and MHRA registered.
Type of Use: Prescription use
Compatible collection tubes available:
Tasso+ Kit Brochure


What is the regulatory status of the Tasso+ Kit?
Tasso+ Kits are procedure packs per Article 22 of the MDR (EU) 2017/745. The Tasso+ device is CE marked and MHRA registered.
What kind of samples does the Tasso+ collect?
The Tasso+ collects liquid samples of whole capillary blood.
How much blood does the Tasso+ collect?
Blood volume is dependent on the type of tube used but averages 200-600μL. Please refer to the tube specifications.
What tubes are compatible with the Tasso+ device?
Refer to the Tasso+ kit brochure for the most up to date kit configurations available. Sample processing instructions will mirror tube manufacturer's labeling.